An R package implementing functions to assist in generating and plotting postestimation quantities after estimating Bayesian regression models using MCMC.
Introduction
BayesPostEst contains functions to generate postestimation quantities after estimating Bayesian regression models. The package was inspired by a set of functions written originally for Johannes Karreth’s workshop on Bayesian modeling at the ICPSR Summer program. It has grown to include new functions (see mcmcReg) and will continue to grow to support Bayesian postestimation. For now, the package focuses mostly on generalized linear regression models for binary outcomes (logistic and probit regression).
Installation
To install the latest release on CRAN:
install.packages("BayesPostEst")The latest development version on GitHub can be installed with:
library("remotes")
install_github("ShanaScogin/BayesPostEst")Once you have installed the package, you can access it by calling:
After the package is loaded, check out the ?BayesPostEst to see a help file.
General setup
Most functions in this package work with posterior distributions of parameters. These distributions need to be converted into a matrix. The mcmcTab function does this automatically for posterior draws generated by JAGS, BUGS, MCMCpack, rstan, and rstanarm. For all other functions, users must convert these objects into a matrix, where rows represent iterations and columns represent parameters. The help file for each function explains how to do this.
Example data
This vignette uses the Cowles dataset (Cowles and Davis 1987, British Journal of Social Psychology 26(2): 97-102) from the carData package.
df <- carData::CowlesThis data frame contains information on 1421 individuals in the following variables:
- neuroticism: scale from Eysenck personality inventory.
- extraversion: scale from Eysenck personality inventory.
- sex: a factor with levels: female; male.
- volunteer: volunteering, a factor with levels: no; yes. This is the outcome variable for the running example in this vignette.
Before proceeding, we convert the two factor variables sex and volunteer into numeric variables. We also means-center and standardize the two continuous variables by dividing each by two standard deviations (Gelman and Hill 2007, Cambridge University Press).
df$female <- (as.numeric(df$sex) - 2) * (-1)
df$volunteer <- as.numeric(df$volunteer) - 1
df$extraversion <- (df$extraversion - mean(df$extraversion)) / (2 * sd(df$extraversion))
df$neuroticism <- (df$neuroticism - mean(df$neuroticism)) / (2 * sd(df$neuroticism))We estimate a Bayesian generalized linear model with the inverse logit link function, where
Pr(Volunteeringi) = logit − 1(β1+β2Femalei+β3Neuroticismi+β4Extraversioni)
BayesPostEst functions accommodate GLM estimates for both logit and probit link functions. The examples proceed with the logit link function. If we had estimated a probit regression, the corresponding argument link in relevant function calls would need to be set to link = "probit". Otherwise, it is set to link = "logit" by default.
Model estimation
To use BayesPostEst, we first estimate a Bayesian regression model. The vignette demonstrates five tools for doing so: JAGS (via the R2jags and rjags packages), MCMCpack, and the two Stan interfaces rstan and rstanarm.
JAGS
First, we prepare the data for JAGS (Plummer 2017). Users need to combine all variables into a list and specify any other elements, like in this case N, the number of observations.
We then write the JAGS model into the working directory.
mod.jags <- paste("
model {
for (i in 1:N){
volunteer[i] ~ dbern(p[i])
logit(p[i]) <- mu[i]
mu[i] <- b[1] + b[2] * female[i] + b[3] * neuroticism[i] + b[4] * extraversion[i]
}
for(j in 1:4){
b[j] ~ dnorm(0, 0.1)
}
}
")
writeLines(mod.jags, "mod.jags") We then define the parameters for which we wish to retain posterior distributions and provide starting values.
params.jags <- c("b")
inits1.jags <- list("b" = rep(0, 4))
inits.jags <- list(inits1.jags, inits1.jags, inits1.jags, inits1.jags)Now, fit the model.
library("R2jags")
set.seed(123)
fit.jags <- jags(data = dl, inits = inits.jags,
parameters.to.save = params.jags, n.chains = 4, n.iter = 2000,
n.burnin = 1000, model.file = "mod.jags")## Compiling model graph
## Resolving undeclared variables
## Allocating nodes
## Graph information:
## Observed stochastic nodes: 1421
## Unobserved stochastic nodes: 4
## Total graph size: 6870
##
## Initializing modelThe same data and model can be used to fit the model using the rjags package:
library("rjags")
mod.rjags <- jags.model(file = "mod.jags", data = dl, inits = inits.jags,
n.chains = 4, n.adapt = 1000)## Compiling model graph
## Resolving undeclared variables
## Allocating nodes
## Graph information:
## Observed stochastic nodes: 1421
## Unobserved stochastic nodes: 4
## Total graph size: 6870
##
## Initializing model
fit.rjags <- coda.samples(model = mod.rjags,
variable.names = params.jags,
n.iter = 2000)MCMCpack
We estimate the same model using MCMCpack (Martin, Quinn, and Park 2011, Journal of Statistical Software 42(9): 1-22).
library("MCMCpack")
fit.MCMCpack <- MCMClogit(volunteer ~ female + neuroticism + extraversion,
data = df, burning = 1000, mcmc = 2000, seed = 123,
b0 = 0, B0 = 0.1)RStan
We write the same model in Stan language.
mod.stan <- paste("
data {
int<lower=0> N;
int<lower=0,upper=1> volunteer[N];
vector[N] female;
vector[N] neuroticism;
vector[N] extraversion;
}
parameters {
vector[4] b;
}
model {
volunteer ~ bernoulli_logit(b[1] + b[2] * female + b[3] * neuroticism + b[4] * extraversion);
for(i in 1:4){
b[i] ~ normal(0, 3);
}
}
")
writeLines(mod.stan, "mod.stan") We then load rstan…
library("rstan")
rstan_options(auto_write = TRUE)
options(mc.cores = parallel::detectCores())… and estimate the model, re-using the data in list format created for JAGS earlier.
rstanarm
Lastly, we use the rstanarm interface (Goodrich, Gabry, Ali, and Brilleman 2019) to estimate the same model again.
Tables of regression coefficients and other parameters
mcmcTab generates a table summarizing the posterior distributions of all parameters contained in the model object. This table can then be used to summarize parameter quantities. The function can directly process objects created by the following packages: R2jags, runjags, rjags, R2WinBUGS, MCMCpack, rstan, rstanarm. This includes the following object classes: jags, rjags, bugs, mcmc, mcmc.list, stanreg, stanfit. By default, mcmcTab generates a dataframe with one row per parameter and columns containing the median, standard deviation, and 95% credible interval of each parameter’s posterior distribution.
mcmcTab(fit.jags)## Variable Median SD Lower Upper
## 1 b[1] -0.455 0.082 -0.615 -0.294
## 2 b[2] 0.232 0.111 0.008 0.442
## 3 b[3] 0.063 0.111 -0.149 0.285
## 4 b[4] 0.514 0.111 0.288 0.733
## 5 deviance 1909.375 2.856 1906.534 1917.190
mcmcTab(fit.rjags)## Variable Median SD Lower Upper
## 1 b[1] -0.460 0.082 -0.622 -0.298
## 2 b[2] 0.233 0.110 0.018 0.449
## 3 b[3] 0.063 0.111 -0.156 0.275
## 4 b[4] 0.520 0.113 0.301 0.746
mcmcTab(fit.MCMCpack)## Variable Median SD Lower Upper
## 1 (Intercept) -0.463 0.083 -0.612 -0.304
## 2 female 0.231 0.110 0.016 0.435
## 3 neuroticism 0.058 0.112 -0.147 0.321
## 4 extraversion 0.509 0.102 0.320 0.718
mcmcTab(fit.stan)## Variable Median SD Lower Upper
## 1 b[1] -0.460 0.081 -0.620 -0.304
## 2 b[2] 0.238 0.110 0.026 0.454
## 3 b[3] 0.058 0.115 -0.167 0.286
## 4 b[4] 0.517 0.111 0.301 0.736
## 5 lp__ -954.790 1.396 -958.640 -953.302
mcmcTab(fit.rstanarm)## Variable Median SD Lower Upper
## 1 (Intercept) -0.459 0.084 -0.624 -0.293
## 2 female 0.236 0.111 0.014 0.449
## 3 neuroticism 0.065 0.111 -0.149 0.280
## 4 extraversion 0.521 0.109 0.306 0.730Proportion of positive/negative draws
Users can add a column to the table that calculates the percent of posterior draws that have the same sign as the median of the posterior distribution.
mcmcTab(fit.jags, Pr = TRUE)ROPE
Users can also define a “region of practical equivalence” (ROPE; Kruschke 2013, Journal of Experimental Psychology 143(2): 573-603). This region is a band of values around 0 that are “practically equivalent” to 0 or no effect. For this to be useful, all parameters (e.g. regression coefficients) must be on the same scale because mcmcTab accepts only one definition of ROPE for all parameters. Users can standardize regression coefficients to achieve this. Because we standardized variables earlier, the coefficients (except the intercept) are on a similar scale and we define the ROPE to be between -0.1 and 0.1.
## This table contains an estimate for parameter values outside of the region of
## practical equivalence (ROPE). For this quantity to be meaningful, all parameters
## must be on the same scale (e.g. standardized coefficients or first differences).
## Variable Median SD Lower Upper PrOutROPE
## 1 b[2] 0.232 0.111 0.008 0.442 0.879
## 2 b[3] 0.063 0.111 -0.149 0.285 0.376
## 3 b[4] 0.514 0.111 0.288 0.733 1.000Conventional regression tables
The mcmcReg function serves as an interface to texreg and produces more polished and publication-ready tables than mcmcTab in HTML or LaTeX format. mcmcReg can produce tables with multiple models with each model in a column and supports flexible renaming of parameters. However, these tables are more similar to standard frequentist regression tables, so they do not have a way to incorporate the percent of posterior draws that have the same sign as the median of the posterior distribution or a ROPE like mcmcTab is able to. Uncertainty intervals can be either standard credible intervals or highest posterior density intervals (Kruschke 2015) using the hpdi argument, and their level can be set with the ci argument (default 95%). Separately calculated goodness of fit statistics can be included with the gof argument.
mcmcReg(fit.jags, format = 'html', doctype = F)Limiting output
mcmcReg supports limiting the parameters included in the table via the pars argument. By default, all parameters saved in the model object will be included. In the case of fit.jags, this include the deviance estimate. If we wish to exclude it, we can specify pars = 'b' which will capture b[1]-b[4] using regular expression matching.
mcmcReg(fit.jags, pars = 'b', format = 'html', doctype = F)If we only wish to exclude the intercept, we can do this by explicitly specifying the parameters we wish to include as a vector. Note that in this example we have to escape the []s in pars because they are a reserved character in regular expressions.
mcmcReg also supports partial regular expression matching of multiple parameter family names as demonstrated below.
Custom coefficient names
mcmcReg supports custom coefficient names to support publication-ready tables. The simplest option is via the coefnames argument. Note that the number of parameters and the number of custom coefficient names must match, so it is a good idea to use pars in tandem with coefnames.
mcmcReg(fit.jags, pars = 'b',
coefnames = c('(Constant)', 'Female', 'Neuroticism', 'Extraversion'),
format = 'html', doctype = F)A more flexible way to include custom coefficient names is via the custom.coef.map argument, which accepts a named list, with names as parameter names in the model and values as the custom coefficient names.
mcmcReg(fit.jags, pars = 'b',
custom.coef.map = list('b[1]' = '(Constant)',
'b[2]' = 'Female',
'b[3]' = 'Nueroticism',
'b[4]' = 'Extraversion'),
format = 'html', doctype = F)The advantage of custom.coef.map is that it can flexibly reorder and omit coefficients from the table based on their positions within the list. Notice in the code below that deviance does not have to be included in pars because its absence from custom.coef.map omits it from the resulting table.
mcmcReg(fit.jags,
custom.coef.map = list('b[2]' = 'Female',
'b[4]' = 'Extraversion',
'b[1]' = '(Constant)'),
format = 'html', doctype = F)However, it is important to remember that mcmcReg will look for the parameter names in the model object, so be sure to inspect it for the correct parameter names. This is important because stan_glm will produce a model object with variable names instead of indexed parameter names.
Multiple models
mcmcReg accepts multiple model objects and will produce a table with one model per column. To produce a table from multiple models, pass a list of models as the mod argument to mcmcReg.
Note, however, that all model objects must be of the same class, so it is not possible to generate a table from a jags object and a stanfit object.
When including multiple models, supplying scalars or vectors to arguments will result in them being applied to each model equally. Treating models differentially is possible by supplying a list of scalars or vectors instead.
Texreg arguments
Although custom.coef.map is not an argument to mcmcReg, it works because mcmcReg supports all standard texreg arguments (a few have been overridden, but they are explicit arguments to mcmcReg). This introduces a high level of control over the output of mcmcReg, as e.g. models can be renamed.
mcmcReg(fit.rstanarm, custom.model.names = 'Binary Outcome', format = 'html', doctype = F)Predicted probabilities
mcmcAveProb
To evaluate the relationship between covariates and a binary outcome, this function calculates the predicted probability (Pr(y = 1)) at pre-defined values of one covariate of interest (x), while all other covariates are held at a “typical” value. This follows suggestions outlined in King, Tomz, and Wittenberg (2000, American Journal of Political Science 44(2): 347-361) and elsewhere, which are commonly adopted by users of GLMs. The mcmcAveProb function by default calculates the median value of all covariates other than x as “typical” values.
Before moving on, we show how create a matrix of posterior draws of coefficients to pass onto these functions. Eventually, each function will contain code similar to the first section of mcmcTab to do this as part of the function.
mcmcmat.jags <- as.matrix(coda::as.mcmc(fit.jags))
mcmcmat.MCMCpack <- as.matrix(fit.MCMCpack)
mcmcmat.stan <- as.matrix(fit.stan)
mcmcmat.rstanarm <- as.matrix(fit.rstanarm)Next, we generate the model matrix to pass on to the function. A model matrix contains as many columns as estimated regression coefficients. The first column is a vector of 1s (corresponding to the intercept); the remaining columns are the observed values of covariates in the model. Note: the order of columns in the model matrix must correspond to the order of columns in the matrix of posterior draws.
mm <- model.matrix(volunteer ~ female + neuroticism + extraversion,
data = df)We can now generate predicted probabilities for different values of a covariate of interest.
Sex
First, we generate full posterior distributions of the predicted probability of volunteering for a typical female and a typical male. In this function and mcmcObsProb, users specify the range of x (here 0 and 1) as well as the number of the column of x in the matrix of posterior draws as well as the model matrix.
aveprob.female.jags <- mcmcAveProb(modelmatrix = mm,
mcmcout = mcmcmat.jags[, 1:ncol(mm)],
xcol = 2,
xrange = c(0, 1),
link = "logit",
ci = c(0.025, 0.975),
fullsims = TRUE)Users can then visualize this posterior distribution using the ggplot2 and ggridges packages.
library("ggplot2")
library("ggridges")
ggplot(data = aveprob.female.jags,
aes(y = factor(x), x = pp)) +
stat_density_ridges(quantile_lines = TRUE,
quantiles = c(0.025, 0.5, 0.975), vline_color = "white") +
scale_y_discrete(labels = c("Male", "Female")) +
ylab("") +
xlab("Estimated probability of volunteering") +
labs(title = "Probability based on average-case approach") +
theme_minimal()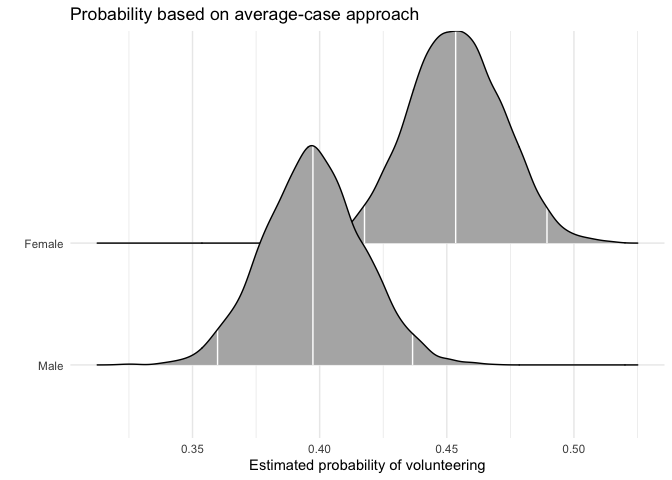
Extraversion
For continuous variables of interest, users may want to set fullsims = FALSE to obtain the median predicted probability along the range of x as well as a lower and upper bound of choice (here, the 95% credible interval).
aveprob.extra.jags <- mcmcAveProb(modelmatrix = mm,
mcmcout = mcmcmat.jags[, 1:ncol(mm)],
xcol = 4,
xrange = seq(min(df$extraversion), max(df$extraversion), length.out = 20),
link = "logit",
ci = c(0.025, 0.975),
fullsims = FALSE)Users can then plot the resulting probabilities using any plotting functions, such as ggplot2.
ggplot(data = aveprob.extra.jags,
aes(x = x, y = median_pp)) +
geom_ribbon(aes(ymin = lower_pp, ymax = upper_pp), fill = "gray") +
geom_line() +
xlab("Extraversion") +
ylab("Estimated probability of volunteering") +
ylim(0, 1) +
labs(title = "Probability based on average-case approach") +
theme_minimal()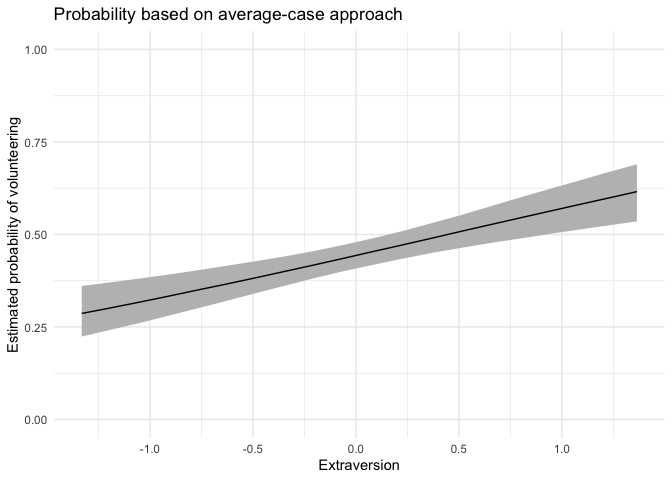
mcmcObsProb
As an alternative to probabilities for “typical” cases, Hanmer and Kalkan (2013, American Journal of Political Science 57(1): 263-277) suggest to calculate predicted probabilities for all observed cases and then derive an “average effect”. In their words, the goal of this postestimation “is to obtain an estimate of the average effect in the population … rather than seeking to understand the effect for the average case.”
Sex
We first calculate the average “effect” of sex on volunteering, again generating a full posterior distribution. Again, xcol represents the position of the covariate of interest, and xrange specifies the values for which Pr(y = 1) is to be calculated.
obsprob.female.jags <- mcmcObsProb(modelmatrix = mm,
mcmcout = mcmcmat.jags[, 1:ncol(mm)],
xcol = 2,
xrange = c(0, 1),
link = "logit",
ci = c(0.025, 0.975),
fullsims = TRUE)Users can again plot the resulting densities.
ggplot(data = obsprob.female.jags,
aes(y = factor(x), x = pp)) +
stat_density_ridges(quantile_lines = TRUE,
quantiles = c(0.025, 0.5, 0.975), vline_color = "white") +
scale_y_discrete(labels = c("Male", "Female")) +
ylab("") +
xlab("Estimated probability of volunteering") +
labs(title = "Probability based on observed-case approach") +
theme_minimal()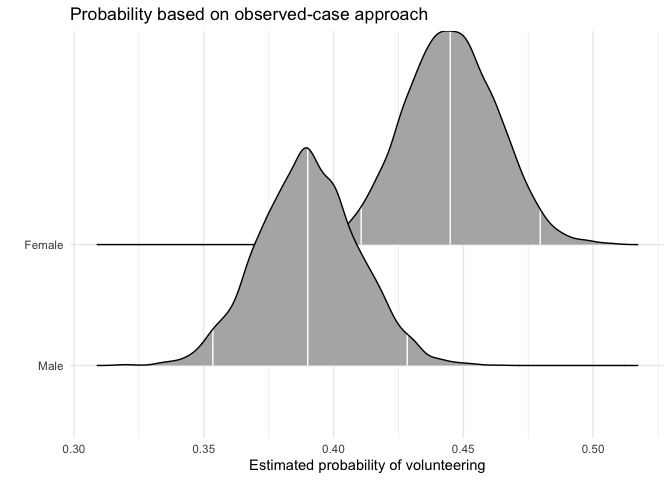
Extraversion
For this continuous predictor, we use fullsims = FALSE.
obsprob.extra.jags <- mcmcObsProb(modelmatrix = mm,
mcmcout = mcmcmat.jags[, 1:ncol(mm)],
xcol = 4,
xrange = seq(min(df$extraversion), max(df$extraversion), length.out = 20),
link = "logit",
ci = c(0.025, 0.975),
fullsims = FALSE)We then plot the resulting probabilities across observed cases.
ggplot(data = obsprob.extra.jags,
aes(x = x, y = median_pp)) +
geom_ribbon(aes(ymin = lower_pp, ymax = upper_pp), fill = "gray") +
geom_line() +
xlab("Extraversion") +
ylab("Estimated probability of volunteering") +
ylim(0, 1) +
labs(title = "Probability based on observed-case approach") +
theme_minimal()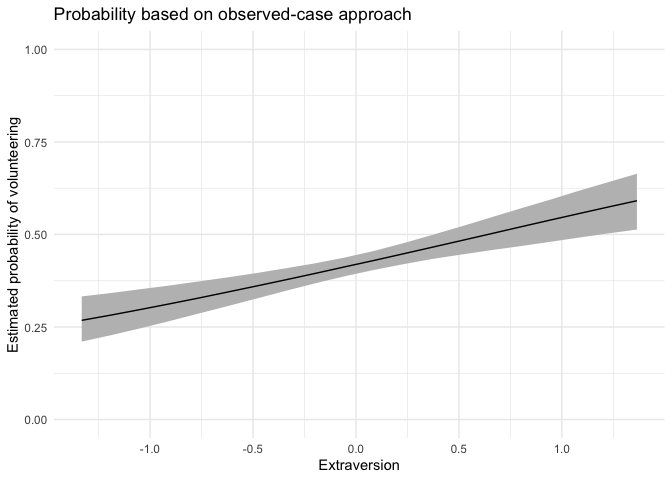
First differences
mcmcFD
To summarize typical effects across covariates, we generate “first differences” (Long 1997, Sage Publications; King, Tomz, and Wittenberg 2000, American Journal of Political Science 44(2): 347-361). This quantity represents, for each covariate, the difference in predicted probabilities for cases with low and high values of the respective covariate. For each of these differences, all other variables are held constant at their median.
fdfull.jags <- mcmcFD(modelmatrix = mm,
mcmcout = mcmcmat.jags[, 1:ncol(mm)],
link = "logit",
ci = c(0.025, 0.975),
fullsims = TRUE)
summary(fdfull.jags)## female neuroticism extraversion
## Min. :-0.04199 Min. :-0.068548 Min. :0.01851
## 1st Qu.: 0.03792 1st Qu.:-0.001534 1st Qu.:0.06982
## Median : 0.05654 Median : 0.011222 Median :0.08131
## Mean : 0.05597 Mean : 0.011369 Mean :0.08109
## 3rd Qu.: 0.07436 3rd Qu.: 0.024294 3rd Qu.:0.09279
## Max. : 0.14808 Max. : 0.090988 Max. :0.13829The posterior distribution can be summarized as above, or users can directly obtain a summary when setting fullsims to FALSE.
fdsum.jags <- mcmcFD(modelmatrix = mm,
mcmcout = mcmcmat.jags[, 1:ncol(mm)],
link = "logit",
ci = c(0.025, 0.975),
fullsims = FALSE)
fdsum.jags## median_fd lower_fd upper_fd VarName VarID
## female 0.05653772 0.001903377 0.10752879 female 1
## neuroticism 0.01122227 -0.026638167 0.05025822 neuroticism 2
## extraversion 0.08131182 0.045476273 0.11589552 extraversion 3Users can plot the median and credible intervals of the summary of the first differences.
ggplot(data = fdsum.jags,
aes(x = median_fd, y = VarName)) +
geom_point() +
geom_segment(aes(x = lower_fd, xend = upper_fd, yend = VarName)) +
geom_vline(xintercept = 0) +
xlab("Change in Pr(Volunteering)") +
ylab("") +
theme_minimal()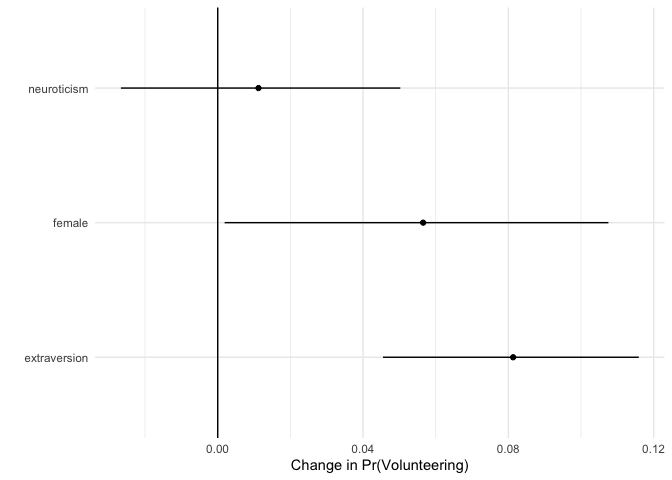
Plotting mcmcFD objects
To make use of the full posterior distribution of first differences, we provide a dedicated plotting method, plot.mcmcFD, which returns a ggplot2 object that can be further customized. The function is modeled after Figure 1 in Karreth (2018, International Interactions 44(3): 463-490). Users can specify a region of practical equivalence and print the percent of posterior draws to the right or left of the ROPE. If ROPE is not specified, the figure automatically prints the percent of posterior draws to the left or right of 0.
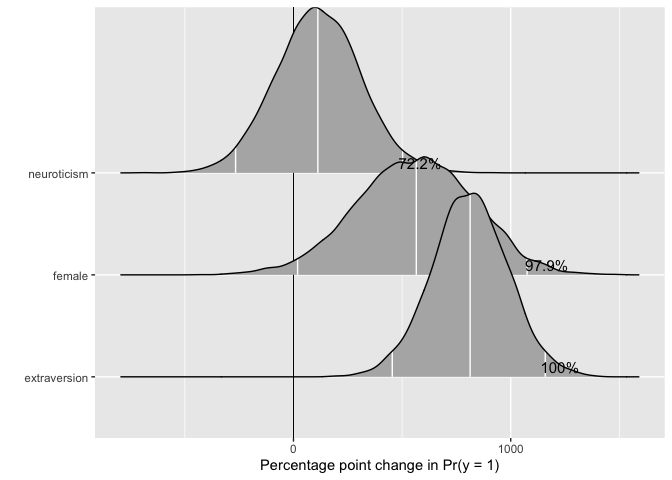
The user can further customize the plot.
p <- plot(fdfull.jags, ROPE = c(-0.01, 0.01))
p + labs(title = "First differences") +
ggridges::theme_ridges()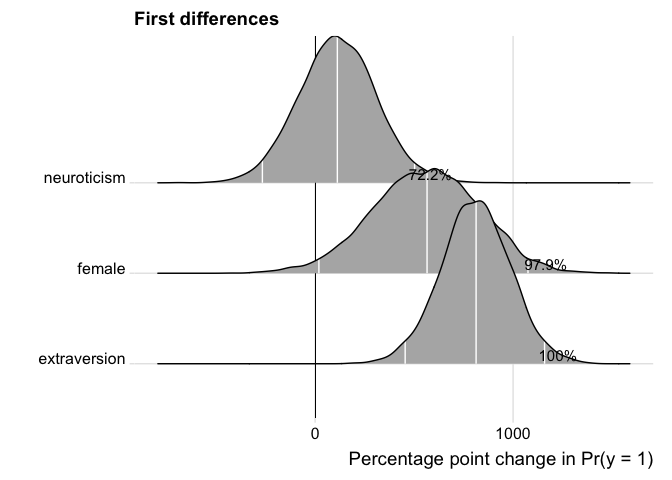
Model fit
mcmcRocPrc
One way to assess model fit is to calculate the area under the Receiver Operating Characteristic (ROC) and Precision-Recall curves. A short description of these curves and their utility for model assessment is provided in Beger (2016). The mcmcRocPrc function produces an object with four elements: the area under the ROC curve, the area under the PR curve, and two dataframes to plot each curve. When fullsims is set to FALSE, the elements represent the median of the posterior distribution of each quantity.
mcmcRocPrc currently requires an “rjags” object (a model fitted in R2jags) as input. Future package versions will generalize this input to allow for model objects fit with any of the other packages used in BayesPostEst.
fitstats <- mcmcRocPrc(object = fit.jags,
yname = "volunteer",
xnames = c("female", "neuroticism", "extraversion"),
curves = TRUE,
fullsims = FALSE)Users can then print the area under the each curve:
fitstats$area_under_roc
fitstats$area_under_prcUsers can also plot the ROC curve…
ggplot(data = as.data.frame(fitstats, what = "roc"), aes(x = x, y = y)) +
geom_line() +
geom_abline(intercept = 0, slope = 1, color = "gray") +
labs(title = "ROC curve") +
xlab("1 - Specificity") +
ylab("Sensitivity") +
theme_minimal()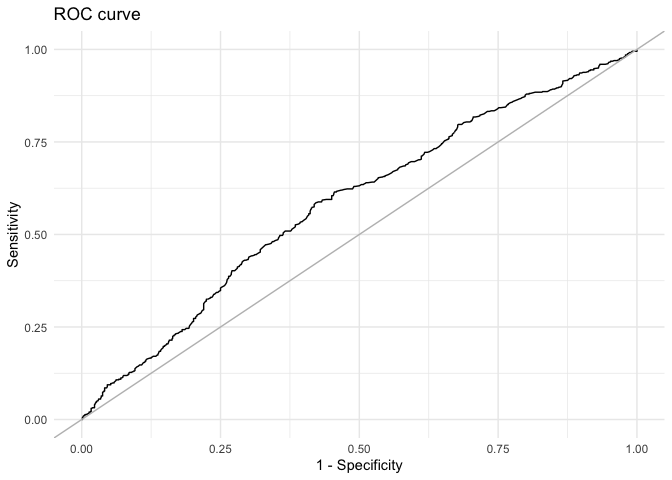
… as well as the precision-recall curve.
ggplot(data = as.data.frame(fitstats, what = "prc"), aes(x = x, y = y)) +
geom_line() +
labs(title = "Precision-Recall curve") +
xlab("Recall") +
ylab("Precision") +
theme_minimal()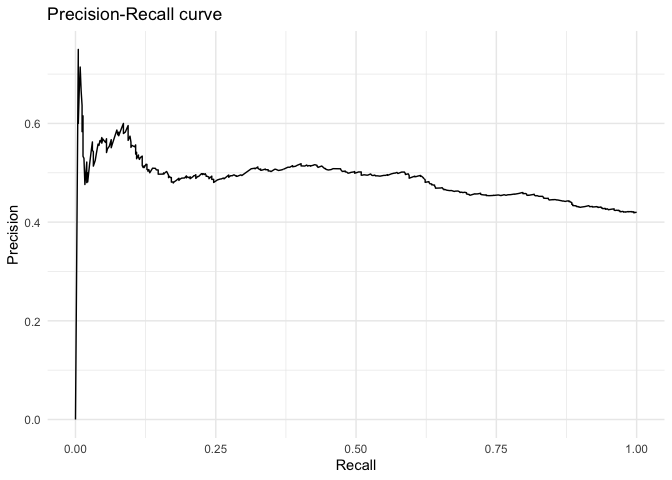
To plot the posterior distribution of the area under the curves, users set the fullsims argument to TRUE. Unless a user wishes to plot credible intervals around the ROC and PR curves themselves, we recommend keeping curves at FALSE to avoid long computation time.
fitstats.fullsims <- mcmcRocPrc(object = fit.jags,
yname = "volunteer",
xnames = c("female", "neuroticism", "extraversion"),
curves = FALSE,
fullsims = TRUE)We can then plot the posterior density of the area under each curve.
ggplot(as.data.frame(fitstats.fullsims),
aes(x = area_under_roc)) +
geom_density() +
labs(title = "Area under the ROC curve") +
theme_minimal()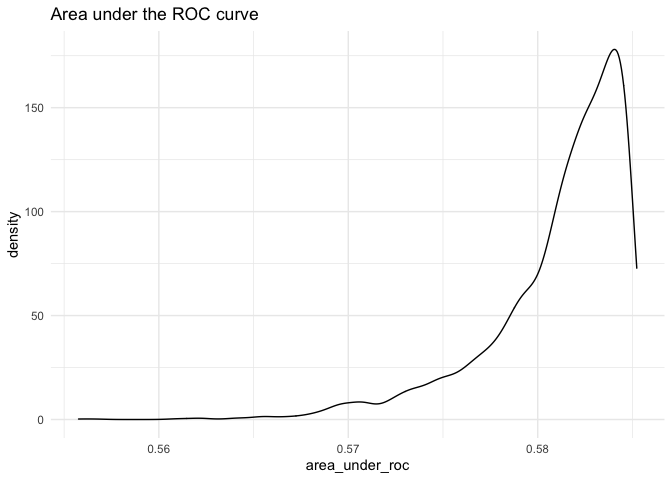
ggplot(as.data.frame(fitstats.fullsims),
aes(x = area_under_prc)) +
geom_density() +
labs(title = "Area under the Precision-Recall curve") +
theme_minimal()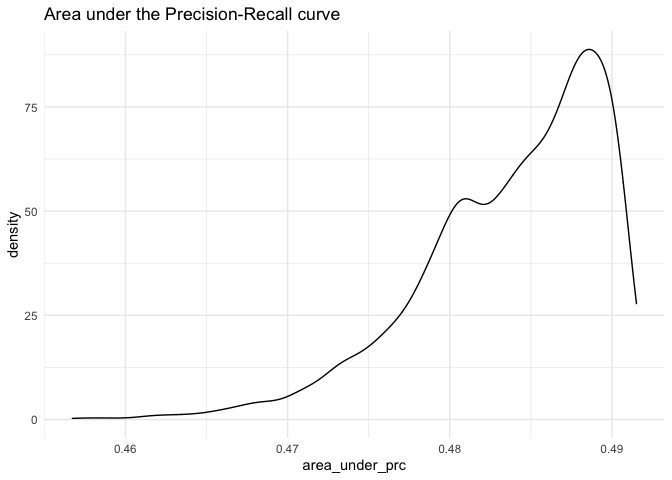
What’s Happening
New functions and enhancements to current functions are in the works. Feel free to browse the issues to see what we are working on or submit an enhancement issue of your own. Our contributing document has more information on ways to contribute.
Contact
Please submit an issue if you encounter any bugs or problems with the package. Feel free to check out Johannes Karreth’s website for more resources on Bayesian estimation.
References
Beger, Andreas. 2016. “Precision-Recall Curves.” Available at: https://doi.org/10.2139/ssrn.2765419.
Cowles, Michael, and Caroline Davis. 1987. “The Subject Matter of Psychology: Volunteers.” British Journal of Social Psychology 26 (2): 97–102. https://doi.org/10.1111/j.2044-8309.1987.tb00769.x.
Fox, John, Sanford Weisberg, and Brad Price. 2018. CarData: Companion to Applied Regression Data Sets. https://CRAN.R-project.org/package=carData.
Gelman, Andrew, and Jennifer Hill. 2007. Data Analysis Using Regression and Multilevel/Hierarchical Models. New York, NY: Cambridge University Press.
Goodrich, Ben, Jonah Gabry, Imad Ali, and Sam Brilleman. 2019. Rstanarm: Bayesian Applied Regression Modeling via Stan. https://mc-stan.org/.
Hanmer, Michael J., and Kerem Ozan Kalkan. 2013. “Behind the Curve: Clarifying the Best Approach to Calculating Predicted Probabilities and Marginal Effects from Limited Dependent Variable Models.” American Journal of Political Science 57 (1): 263–77. https://doi.org/10.1111/j.1540-5907.2012.00602.x.
Karreth, Johannes. 2018. “The Economic Leverage of International Organizations in Interstate Disputes.” International Interactions 44 (3): 463–90. https://doi.org/10.1080/03050629.2018.1389728.
King, Gary, Michael Tomz, and Jason Wittenberg. 2000. “Making the Most of Statistical Analyses: Improving Interpretation and Presentation.” American Journal of Political Science 44 (2): 347–61. https://doi.org/10.2307/2669316.
Kruschke, John K. 2013. “Bayesian Estimation Supersedes the T-Test.” Journal of Experimental Psychology: General 142 (2): 573–603. https://doi.org/10.1037/a0029146.
Kruschke, John K. 2015. Doing Bayesian Data Analysis: A Tutorial with R, JAGS, and Stan. Amsterdam: Academic Press. 978-0-12-405888-0
Long, J. Scott. 1997. Regression Models for Categorical and Limited Dependent Variables. Thousand Oaks: Sage Publications.
Martin, Andrew D., Kevin M. Quinn, and Jong Hee Park. 2011. “MCMCpack: Markov Chain Monte Carlo in R.” Journal of Statistical Software 42 (9):
Plummer, Martyn. 2017. “JAGS Version 4.3.0 User Manual.” https://mcmc-jags.sourceforge.io/.
Stan Development Team. 2019. RStan: The R Interface to Stan. https://mc-stan.org/.